$\require{mhchem}$
Properties of Hydrates
===
## Introduction
Ionic compounds are created through the formation of ionic bonds between metal and nonmetal elements. Most ionic compounds are pure compounds. Some of these ionic compounds may absorb water in humid conditions and lose water in dry conditions. An example of this is sodium chloride (table salt). In humid conditions, it may start to "clump" together. Other ionic compounds contain water as part of their crystal lattice. These compounds are referred to as **hydrates**. Hydrates contain a fixed number of water molecules per molecule of compound. An example is barium hydroxide octahydrate.
$$
\ce{Ba(OH)2.8H2O}
$$
This formula shows that for every one mole of Ba(OH)~2~ there are 8 moles of water present. You can determine the mass% of water present in a hydrate using equation 1.
$$
\%\text{Mass} \text{ Water} =
\frac{
\text{Mass of Water}
}
{
\text{Mass of one Mole of Hydrated Compound}
}
\times
100\%
$$
Since the molar mass of $\ce{Ba(OH)2.8H2O}$ is 315.3 $\frac{\textsf{g}}{\textsf{mol}}$ and the mass of the 8 moles of water is
$$
8 \, \textsf{mol} \left( \frac{18 \, \textsf{g}}{\textsf{mol}} \right) = 144 \, \textsf{g} \, \text{water}
$$
the %mass of Water in $\ce{Ba(OH)2.8H2O}$ would be
$$
\% \text{Mass} \, \text{Water} = \frac{144 \, \textsf{g} \, \text{Water}}{315.3 \, \textsf{g} \, \ce{Ba(OH)2}}
\times
100\%
= 45.67\%
$$
Hydrates lose their incorporated water molecules when they are heated above the boiling point of water. The dried form of a hydrate is called an **anhydride**.
$$
\ce{Ba(OH)2 ($s$).8 H2O ->[Heat] Ba(OH)2 ($s$) + 8 H2O ($g$)}
$$
You can determine the mass of water lost by using equation 6.
$$
\%\text{Mass Lost} =
\frac{\text{Mass of Hydrate - Mass of Anhydride}}{\text{Mass of Hydrate}}
\times
100\%
$$
Generally, there are three rules when determining whether a compound is a hydrate.
1. The compound loses water upon heating
2. The process is reversible i.e. when water is added it is reincorporated into the compound's lattice.
3. If there was a color change when the hydrate was dried, the compound returns to the original color when water is reincorporated into the ionic lattice.
In this experiment, you will be studying four compounds to determine whether they are hydrates or not. You will also be determining the formula of a hydrate by calculating the amount of water that is lost.
### Sample problem
You suspect that a sample of tin(IV) chloride is a hydrate. You weigh out 1.000 g of the green solid and place it in a crucible. After thoroughly heating the sample over a Bunsen burner the solid turns blue. You reweigh the sample and determine that 0.878 g of blue solid remains. You add the blue solid to a small portion of water in a beaker. The solid dissolves completely to yield a green solution. What is the %mass lost by the tin(IV) chloride? Is it a hydrate? If so, what is the formula of the hydrate?
We can calculate the %mass lost by using equation 2.
$$
\% \text{Mass Lost} = \frac{1.000 \, \textsf{g} - 0.878 \, \textsf{g}}{1.000 \, \textsf{g}}
\times
100\%
= 12.2\%
$$
In order to determine the formula of the hydrate, we will need two additional pieces of information. First we will need to determine the formula of anhydrous tin(IV) chloride. Since tin is labeled in its name as having a 4+ charge and chlorine ions have a charge of 1-, the formula of anhydrous tin(IV) chloride is $\ce{SnCl4}$. Next, we will need to know the molar mass of anhydrous tin(IV) chloride. This can be determined by summing the atomic masses of tin and chlorine present in $\ce{SnCl4}$.
$$
1 \left( 118.71 \, \frac{\textsf{g}}{\textsf{mol} \, \ce{Sn}} \right) +
4 \left( 35.45 \, \frac{\textsf{g}}{\textsf{mol} \, \ce{Cl}} \right) = 260.5 \, \frac{\textsf{g}}{\textsf{mol} \, \ce{SnCl4}}
$$
By dividing that mass of the anhydride by the molar mass, we can determine the amount of moles of tin (IV) chloride present.
$$
0.878 \, \textsf{g} \left( \frac{\textsf{mol} \, \ce{SnCl4}}{ 260.5 \, \textsf{g}} \right) = 3.37 \times 10^{-3} \hspace{2mm} \textsf{mol} \, \ce{SnCl4}
$$
The remaining mass (the mass of hydrate minus the mass of the anhydride) is the mass of the water. This can be divided by the molar mass of water $\left( 18.02 \ce{\frac{g}{mol}} \, \right)$to determine the moles of water present.
$$
\frac{1.000 \, \textsf{g} - 0.878 \, \textsf{g}}{18.02 \hspace{1mm} \frac{\textsf{g}}{\textsf{mol}}} = \frac{0.122 \, \textsf{g}} { 18.02 \, \frac{\textsf{g}}{\textsf{mol}}} = 6.77 \times 10^{-3} \, \textsf{mol}\, \text{water}
$$
Finally, determine the ratio of water to $\ce{SnCl4}$.
$$
\frac{ 6.77 \times 10^{-3} \hspace{2mm} \textsf{mol} \, \text{water} }{ 3.37 \times 10^{-3} \hspace{2mm} \textsf{mol} \, \ce{SnCl4} } = \frac{ 2.01 \, \textsf{mol} \, \text{water} }{ 1 \, \textsf{mol} \, \ce{SnCl4} }
$$
You'll notice that when significant digits are applied to our calculations, we wind up with a water to $\ce{SnCl4}$ ratio that is not a whole number. This is to be expected, we will just round to the nearest whole number and assume that there are two water molecules for each $\ce{SnCl4}$ molecule. The formula for the tin(IV) chloride hydrate is therefore **$\ce{SnCl4.2 H2O}$**.
## Materials
- Ring Stand
- Clay Triangle
- Bunsen burner
- Ring Clamp
- Striker
- Crucible
- Tongs
- Hot Mittens
- Ionic Solid
- Clay Tile
- Desiccator
## Procedure
:::warning
⚠️ There is some inherit danger to heating glass over a Bunsen burner. The glass may shatter or even melt with the intense heat of the flame. To mitigate this risk, chemists often use ceramic vessels when heating on a Bunsen burner. Ceramics are incredibly heat resistant. However, they are porous and absorb water when exposed to the humidity of open air. They must therefore be preheated before use in order to obtain accurate mass measurements.
:::
:::danger
🔥 → 🧯 Using open flame in the lab is very dangerous. Your instructor will demonstrate the correct procedure for lighting and adjusting the flame. Pay careful attention and always be aware of your surroundings. Clear your workspace of flammables and arrange your workspace to avoid tipping over your Bunsen burner.
:::
### Part One: Prepare your crucible
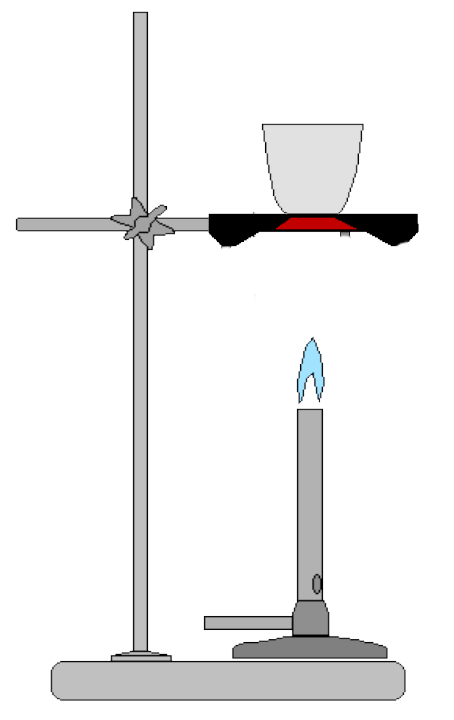
1. Arrange your ring stand, Bunsen burner and clay triangle as shown by your instructor.
1. Light your Bunsen burner and adjust it to a small blue flame.
1. Using your tongs, place your crucible onto the clay triangle and heat it intensely for 3 minutes. If you see a faint red glow, you are finished.
1. Turn off your Bunsen burner and transfer your crucible with your tongs to your clay tile to cool. You cannot *see* how hot your crucible is. You can test the temperature of your crucible by placing the tip of thermometer against it for a few seconds. Do not hold the tip of the thermometer against the crucible for an extended period of time when it is very hot.
1. When your crucible is cool, take it to the balance and determine its tare weight.
### Part Two: Qualitative Analysis of Potential Hydrates
1. Weigh out approximately 0.5 grams of one of the potential hydrates into your crucible.
1. Take the crucible/sample back to your bench and gently heat the sample.
1. Note any steam rising or color change as you heat. You can gently stir the solid to aid in drying.
1. Turn off your burner and allow it cool on your clay tile.
1. After your crucible has cooled, reweigh the sample. Calculate the %mass lost. A %mass lost greater than 5% is strong evidence of a hydrate.
1. Transfer a small amount of the sample to a test tube. Dispose of the rest by scraping it into the appropriate waste container. A small amount of residue left in the crucible is okay.
1. Repeat steps 1 - 6 for another two samples.
1. Lastly, return to the three samples you've collected in test tubes. Add a small portion of water to each tube and note the the color of the solutions they form.
#### Cobalt(II) Sulfate
<iframe width="100%" height="410" src="https://www.youtube.com/embed/wvzNpjxQG6w" title="Chemnotes.org" frameborder="0" allow="accelerometer; autoplay; clipboard-write; encrypted-media; gyroscope; picture-in-picture; web-share" referrerpolicy="strict-origin-when-cross-origin" allowfullscreen></iframe>
##### Initial

##### After Heating

##### Solution
#### Cobalt(II) Chloride
<iframe width="100%" height="410" src="https://www.youtube.com/embed/qp8rvilfN6w" title="Chemnotes.org" frameborder="0" allow="accelerometer; autoplay; clipboard-write; encrypted-media; gyroscope; picture-in-picture; web-share" referrerpolicy="strict-origin-when-cross-origin" allowfullscreen></iframe>
##### Initial

##### After Heating

##### Solution
#### Calcium Carbonate
<iframe width="100%" height="410" src="https://www.youtube.com/embed/wj64XoW3nrU" title="Chemnotes.org" frameborder="0" allow="accelerometer; autoplay; clipboard-write; encrypted-media; gyroscope; picture-in-picture; web-share" referrerpolicy="strict-origin-when-cross-origin" allowfullscreen></iframe>
##### Initial

##### After Heating

##### Solution


### Part Three: Quantitative Analysis of an Unknown Hydrate
:::info
👉 In this part, you will need to be more careful in order to get data that will allow you to accurately determine the formula of your hydrate. If there is significant residue remaining in your crucible, it may be necessary to rinse it out with a small portion of water and then prepare it again as you did in part one. Do not add water to a hot crucible.
:::
1. Obtain about about 1.5 grams of an unknown hydrate from your instructor.
1. Weigh the sample in your crucible.
1. Heat this sample gently in the flame for 10 minutes and intensely for 5 minutes.
1. Let it cool briefly on your clay tile and then transfer it to the desiccator. A desiccator is an air tight chamber that contains desiccant, chemical drying agent that ensures a low humidity environment inside the chamber. Allowing the crucible to cool inside the desiccator will ensure that water is not reabsorbed from the air.
1. Once the crucible is cool, mass the sample and crucible again. Make note of any color change that occurred during the heating process.
#### Copper(II) Sulfate
<iframe width="100%" height="410" src="https://www.youtube.com/embed/wck4x98mjDs" title="Chemnotes.org" frameborder="0" allow="accelerometer; autoplay; clipboard-write; encrypted-media; gyroscope; picture-in-picture; web-share" referrerpolicy="strict-origin-when-cross-origin" allowfullscreen></iframe>

## Data Sheet
{%pdf https://public.chemnotes.org/lab/sheets/propertiesofhydrates.pdf %}