<style>
/* Width of text in slides */
.slides { width: 1200px !important; }
/* line space bullet list */
.reveal .slides section, .reveal .slides section > section { line-height: 1.5; }
/* headers */
.reveal h1 {
margin-bottom: 1em;
font-weight: bold;
font-size: 32pt }
.reveal h2 {
margin-bottom: 1em;
font-weight: bold;
font-size: 30pt
}
.reveal h3 {
margin-bottom: 1em;
font-weight: bold;
font-size: 28pt
}
.reveal h4 {
margin-bottom: 1em;
font-weight: bold;
font-size: 26pt
}
.reveal h5 {
margin-bottom: 1em;
font-weight: bold;
font-size: 24pt
}
.reveal h6 {
margin-bottom: 1em;
font-weight: normal;
font-size: 24pt
}
/* text */
.reveal {
font-size: 24pt;
font-weight: normal;
}
/* logo as background */
.container {
background: no-repeat url(https://www.mcc.edu/MottStrong/images/mott-strong-3c-logo.png) top 20px left 10px; background-size: 5% !important;
} code {color: #000000}
.reveal table th { font-size: 1em}
.reveal table td { font-size: 0.75em}
/* Remove Border arround images */
.reveal section img {
background:none;
border:none;
box-shadow:none;
object-fit: contain;
margin: 0;
padding: 0;
max-width: 100%;
}
.reveal figure img {
box-shadow:none;
background:none;
border:none;
max-width: 100%;
object-fit: contain;
}
</style>
$\require{mhchem}$
OpenStax Chemistry 2e Chapter 10 Section 1
===
Michael Stogsdill
Mott Community College
---
## Learning Objectives
- Describe the types of intermolecular forces possible between atoms or molecules in condensed phases (dispersion forces, dipole-dipole attractions, and hydrogen bonding)
- Identify the types of intermolecular forces experienced by specific molecules based on their structures
- Explain the relation between the intermolecular forces present within a substance and the temperatures associated with changes in its physical state
---
## KMT for Gases
1. Gases are composed of molecules that are in continuous motion, travelling in straight lines and changing direction only when they collide with other molecules or with the walls of a container.
2. The molecules composing the gas are negligibly small compared to the distances between them.
3. The pressure exerted by a gas in a container results from collisions between the gas molecules and the container walls.
4. Gas molecules exert no attractive or repulsive forces on each other or the container walls; therefore, their collisions are elastic (do not involve a loss of energy).
5. The average kinetic energy of the gas molecules is proportional to the kelvin temperature of the gas.
Note:
The nature of gases allows us to make a number of convenient assumptions. In general, they behave predictably. A stoichastic systemm that can be modeled using statistics leading to a rich mathematical model.
---
## KMT for liquids and Solids
- Particles in a solid are tightly packed together and often arranged in a regular pattern; in a liquid, they are close together with no regular arrangement; in a gas, they are far apart with no regular arrangement.
- Particles in a solid vibrate about fixed positions and do not generally move in relation to one another; in a liquid, they move past each other but remain in essentially constant contact; in a gas, they move independently of one another except when they collide.
Note:
The addition of intermolecular forces that vary from system to system makes describing condensed systems more difficult. We will need to use new tools to gather less useful information.
---
## Question 2
In terms of the kinetic molecular theory, in what ways are liquids similar to solids? In what ways are liquids different from solids?
----
**Similarities**
Both are composed of atoms, ions, or molecules; there is little empty space between these particles; the motion of the particles changes with changing temperature.
**Differences**
<!-- .element: class="fragment" data-fragment-index="1" -->
Particles in the solid are fixed in a regular arrangement; although they vibrate, they do not move about; particles in the liquid have no regular arrangement and are free to move past each other.
<!-- .element: class="fragment" data-fragment-index="1" -->
---
## Question 3
In terms of the kinetic molecular theory, in what ways are liquids similar to gases? In what ways are liquids different from gases?
----
<p style="text-align:left; color:green;">They are similar in that the atoms or molecules are free to move from one position to another. They differ in that the particles of a liquid are confined to the shape of the vessel in which they are placed. In contrast, a gas will expand without limit to fill the space into which it is placed.</p>
---
## *Intra*molecular vs. *Inter*molecular Forces
- **Intra**molecular forces are interactions that occur winthin the same molecule.
- **Inter**molecular forces are interactions that occur between two or more molecules.
<img src="https://openstax.org/apps/archive/20241024.164013/resources/031c6194fc6c4b7316455344ac5144bbd91a33a5" alt="An image is shown in which two molecules composed of a green sphere labeled “C l” connected on the right to a white sphere labeled “H” are near one another with a dotted line labeled “Intermolecular force ( weak )” drawn between them. A line connects the two spheres in each molecule and the line is labeled “Intramolecular force ( strong ).”" style="zoom: 67%;" />
---
## Long Range Intramolecular Forces
<img src="https://doc.chemnotes.org/uploads/f54497a9-75f8-4124-afa5-da6e83ff2991.png" alt="Image of 2-hydroxy-benzaldehyde. The hydrogen bonding between the aldehyde group and the alcohol group is highlighted with a dotted orange line." style="zoom: 50%;" />
Note:
Image of 2-hydroxy-benzaldehyde. The hydrogen bonding between the aldehyde group and the alcohol group is highlighted with a dotted orange line.
---
## Networked Intermolecular Forces
<img src="https://doc.chemnotes.org/uploads/7224aa2d-06ef-4052-aed0-fe0c283bcfec.png" alt="Image of three water molecules their hydrogen bonding is highlighted in " style="zoom:67%;" />
Note:
IMFs can arrange themselves into complicated long range networks. The overall effect on bulk properties can be increased by either increasing the strength of individual interactions or increasing the possible number of interactions.
---
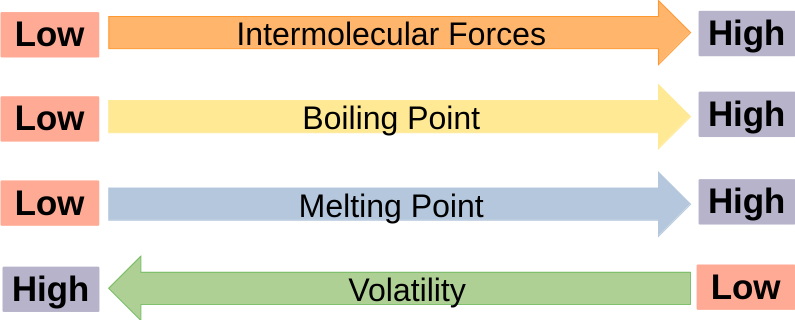
## IMFS Effect on Bulk Properties
---
## Question 1
In terms of their bulk properties, how do liquids and solids differ? How are they similar?
----
<p style="text-align:left; color:green;">Liquids and solids are similar in that they are matter composed of atoms, ions, or molecules. They are incompressible and have similar densities that are both much larger than those of gases. They are different in that liquids have no fixed shape, and solids are rigid.</p>
---
## IMFs Effect on Phase
- The phase in which a substance exists depends on the relative extents of its intermolecular forces (IMFs) and the kinetic energies (KE) of its molecules.
<img src="https://openstax.org/apps/archive/20241024.164013/resources/99c7f7d4b6b2e63894a1a13d4f04329990d6c8ed" alt="Three sealed flasks are labeled, “Crystalline solid,” “Liquid,” and “Gas,” from left to right. The first flask holds a cube composed of small spheres sitting on the bottom while the second flask shows a lot of small spheres in the bottom that are spaced a small distance apart from one another and have lines around them to indicate motion. The third flask shows a few spheres spread far from one another with larger lines to indicate motion. There is a right-facing arrow that spans the top of all three flasks. The arrow is labeled, “Increasing K E ( temperature ).” There is a left-facing arrow that spans the bottom of all three flasks. The arrow is labeled, “Increasing I M F.”" style="zoom:67%;" />
---
## [PHeT States of Matter](https://phet.colorado.edu/sims/html/states-of-matter/latest/states-of-matter_all.html)
Note:
Phase Change -> Select "Adjustable Attraction"
---
## van der Waals Forces
| Interaction | Cause |
| :--------- | :--- |
| Dispersion (van der Waal) | Instantaneous Dipole |
| Dipole - Dipole | Dipole Moments |
| Hydrogen Bonding | H interacting with F, O, or N |
Note:
All of the attractive forces between neutral atoms and molecules are known as van der Waals forces, although they are usually referred to more informally as intermolecular attraction.
---
## Dispersion Forces
- **Instantaneous dipoles** can be induced in any molecule or atom
- This is a weak, non-permanent interaction
- :arrow_up: Strength :arrow_up: Formula Mass :arrow_up: Surface Area
- The degree to which a molecule's charge distribution can be distorted is called **polarizability**
$$
\mu =q\vec{r}
$$
Note:
A molecule that has a charge cloud that is easily distorted is said to be very polarizable and will have large dispersion forces; one with a charge cloud that is difficult to distort is not very polarizable and will have small dispersion forces.
---
<img src="https://doc.chemnotes.org/uploads/161e5162-427a-4fcf-ac45-062099be09db.png" alt="Image showing three atoms before an induced dipole. The electrons are randomly arranged around the nuclei." style="zoom:67%;" />
<img src="https://doc.chemnotes.org/uploads/f841aa96-6eab-4bf2-a27f-566160c709b0.png" alt="Image showing three atoms interacting to induce a dipole. The electrons are arranged asymmetrically around the nuclei." style="zoom:67%;" />
Note:
Because the electrons of an atom or molecule are in constant motion (or, alternatively, the electron’s location is subject to quantum-mechanical variability), at any moment in time, an atom or molecule can develop a temporary, instantaneous dipole if its electrons are distributed asymmetrically. The presence of this dipole can, in turn, distort the electrons of a neighboring atom or molecule, producing an induced dipole. These two rapidly fluctuating, temporary dipoles thus result in a relatively weak electrostatic attraction between the species—a so-called dispersion force
---

---
## Melting and Boiling Points of the Halogens
| Halogen | Molar Mass | Atomic Radius | Melting Point | Boiling Point |
|:--------|:----------:|:-------------:|:-------------:|:-------------:|
| fluorine, F~2~ | 38 g/mol | 72 pm | 53 K | 85 K |
| chlorine, Cl~2~ | 71 g/mol | 99 pm | 172 K | 238 K |
| bromine, Br~2~ | 160 g/mol | 114 pm | 266 K | 332 K |
| iodine, I~2~ | 254 g/mol | 133 pm | 387 K | 457 K |
| astatine, At~2~ | 420 g/mol | 150 pm | 575 K | 610 K |
<strong>Table 10.1</strong>
Note:
The larger the atomic radius (formula mass) the higher the transition tempeeratures.
---
## Surface Area and Dispersion Forces
<img src="https://openstax.org/apps/archive/20241024.164013/resources/0a03d691767b3bbe6caa07a9d6c246346fbbac1d" alt="Three images of molecules are shown. The first shows a cluster of large, gray spheres each bonded together and to several smaller, white spheres. There is a gray, jagged line and then the mirror image of the first cluster of spheres is shown. Above these two clusters is the label, “Small contact area, weakest attraction,” and below is the label, “neopentane boiling point: 9.5 degrees C.” The second shows a chain of three gray spheres bonded by the middle sphere to a fourth gray sphere. Each gray sphere is bonded to several smaller, white spheres. There is a jagged, gray line and then the mirror image of the first chain appears. Above these two chains is the label, “Less surface area, less attraction,” and below is the label, “isopentane boiling point: 27 degrees C.” The third image shows a chain of five gray spheres bonded together and to several smaller, white spheres. There is a jagged gray line and then the mirror image of the first chain appears. Above these chains is the label, “Large contact area, strong attraction,” and below is the label, “n-pentane boiling point 36 degrees C.”" style="zoom: 67%;" />
Note:
The strength of the dispersion forces increases with the contact area between molecules, as demonstrated by the boiling points of these pentane isomers.
---
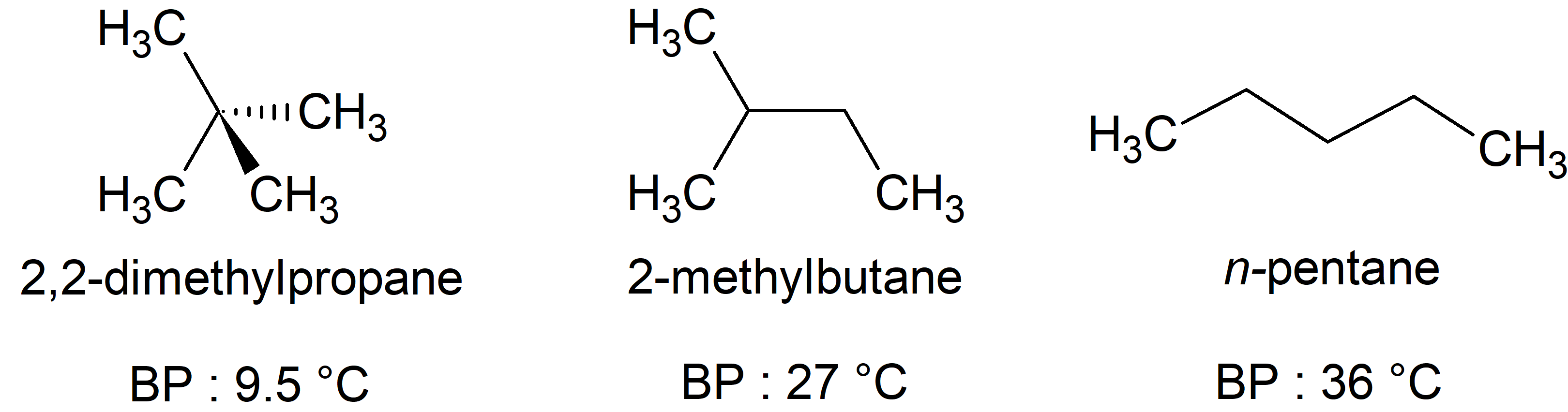
Note:
As predicted, the boiling point increases for less branched constitutional isomers.
---
## Periodic Table of Electronegativity

---
## Dipole - Dipole Interactions
- When there exists a difference of electronegativity across a bond, a **Bond Dipole** will exist.
- This is a permanent *intra*molecular interaction.
- Larger electronegativity differences and longer bonds lead to stronger bond dipoles
- Bond dipoles combine through vector addition to yield **Dipole Moments**.
- These are *inter*molecular forces that effect bulk behavior
- **Dipole-Dipole Attractions** are stronger than Dispersion Forces.
Note:
dipole-dipole attractions are the electrostatic force between the partially positive end of one polar molecule and the partially negative end of another.
---

Note:
This image shows two arrangements of polar molecules, such as HCl, that allow an attraction between the partial negative end of one molecule and the partial positive end of another.
---
## Dipole-Dipole Attraction effect on Bulk Properties
| Compound | Dipole | Formula Mass | Boiling Point |
|:---------|:------:|:------------:|:-------------:|
| Flourine, F~2~ | No | 37.997 g/mol | 85 K |
| Hydrochloric Acid, HCl | Yes | 36.461 g/mol | 188 K |
---
## Dipole-Ion Attraction
- Water and other polar molecules are attracted to ions.
- The electrostatic attraction between an ion and a molecule with a dipole is called an **ion-dipole attraction**.
- These attractions play an important role in the dissolution of ionic compounds in water.
- Dipole-Ion Attractions are typically stronger than Dipole-Dipole Attractions.
---
<img src="https://openstax.org/apps/archive/20241024.164013/resources/a7861ff5f9f64e6c9e179970d0231adf584fd506" alt="The diagram shows eight purple spheres labeled K superscript plus and eight green spheres labeled C l superscript minus mixed and touching near the center of the diagram. Outside of this cluster of spheres are seventeen clusters of three spheres, which include one red and two white spheres. A red sphere in one of these clusters is labeled O. A white sphere is labeled H. Two of the green C l superscript minus spheres are surrounded by three of the red and white clusters, with the red spheres closer to the green spheres than the white spheres. One of the K superscript plus purple spheres is surrounded by four of the red and white clusters. The white spheres of these clusters are closest to the purple spheres." style="zoom:67%;" />
Note:
As potassium chloride (KCl) dissolves in water, the ions are hydrated. The polar water molecules are attracted by the charges on the K^+^ and Cl^−^ ions. Water molecules in front of and behind the ions are not shown.
---
## Hydrogen Bonding
- Hydrogen Bonding is a special case of Dipole-Dipole interactions.
- When hydrogen is bound to a sufficiently electronegative atom (O, N, or F) the single electron on the atom is drawn far away from the nucleus.
- The result is highly concentrated partial charge leading to a stronger than normal interaction.
Note:
Despite use of the word “bond,” keep in mind that hydrogen bonds are intermolecular attractive forces, not intramolecular attractive forces (covalent bonds). Hydrogen bonds are much weaker than covalent bonds, only about 5 to 10% as strong, but are generally much stronger than other dipole-dipole attractions and dispersion forces.
---

Note:
Water molecules participate in multiple hydrogen-bonding interactions with nearby water molecules.
---
## Effect of Hydrogen Bonding on Bulk Properties
| Compound | Dipole |Hydrogen Bonding | Formula Mass | Boiling Point |
|:---------|:------:|:---------------:|:------------:|:-------------:|
| Nitrosyl Fluoride, ONF | Yes | No | 49.005 $\frac{\textsf{g}}{\textsf{mol}}$ | 201 K |
| Water, H~2~O | Yes | Yes | 18.015 $\frac{\textsf{g}}{\textsf{mol}}$ | 373 K |
Note:
Both molecules have about the same shape and ONF is the heavier and larger molecule. It is, therefore, expected to experience more significant dispersion forces. Both molecules are polar and exhibit comparable dipole moments. The large difference between the boiling points is due to a particularly strong hydrogen bonding.
---
## Hydrides
- The boiling points of hydrides in Groups 17, 16, and 15 exhibit a significant break in the trend predicted by considering dispersive forces alone.
<img src="https://openstax.org/apps/archive/20241024.164013/resources/cb929211736bd1033ffd79429811722ae25e128e" alt="A line graph is shown where the y-axis is labeled “Boiling point (, degree sign, C )” and has values of “ negative 150” to “150” from bottom to top in increments of 50. The x-axis is labeled “Period” and has values of “0” to “5” in increments of 1. Three lines are shown on the graph and are labeled in the legend. The red line is labeled as “halogen family,” the blue is “oxygen family” and the green is “nitrogen family.” The first point on the red line is labeled “question mark” and is at point “2, negative 120”. The second point on the line is labeled “H C l” and is at point “3, negative 80” while the third point on the line is labeled “H B r” and is at point “4, negative 60”. The fourth point on the line is labeled “H I” and is at point “5, negative 40.” The first point on the green line is labeled “question mark” and is at point “2, negative 125.” The second point on the line is labeled “P H, subscript 3” and is at point “3, negative 80” while the third point on the line is labeled “A s H, subscript 3” and is at point “4, negative 55.” The fourth point on the line is labeled “S b H, subscript 3” and is at point “5, negative 10.” The first point on the blue line is labeled “question mark” and is at point “2, negative 80.” The second point on the line is labeled “H, subscript 2, S” and is at point “3, negative 55” while the third point on the line is labeled “H, subscript 2, S e” and is at point “4, negative 45.” The fourth point on the line is labeled “H, subscript 2, T e” and is at point “5, negative 3.”" style="zoom:50%;" />
Note:
For the group 15, 16, and 17 hydrides, the boiling points for each class of compounds increase with increasing molecular mass for elements in periods 3, 4, and 5.
---
<img src="https://openstax.org/apps/archive/20241024.164013/resources/b8d6a918b6f044e7b733c5a970036eba02dea041" alt="A line graph is shown where the y-axis is labeled “Boiling point, ( degree sign, C )” and has values of “negative 150” to “150” from bottom to top in increments of 50. The x-axis is labeled “Period” and has values of “0” to “5” in increments of 1. Three lines are shown on the graph and are labeled in the legend. The red line is labeled as “halogen family,” the blue is “oxygen family” and the green is “nitrogen family.” The first point on the red line is labeled “H F” and is at point “2, 25.” The second point on the line is labeled “H C l” and is at point “3, negative 80” while the third point on the line is labeled “H B r” and is at point “4, negative 60.” The fourth point on the line is labeled “H I” and is at point “5, negative 40.” The first point on the green line is labeled “N H, subscript 3” and is at point “2, negative 40.” The second point on the line is labeled “P H, subscript 3” and is at point “3, negative 80” while the third point on the line is labeled “A s H, subscript 3” and is at point “4, negative 55.” The fourth point on the line is labeled “S b H, subscript 3” and is at point “5, negative 10.” The first point on the blue line is labeled “H, subscript 2, O” and is at point “2, 100.” The second point on the line is labeled “H, subscript 2, S” and is at point “3, negative 55” while the third point on the line is labeled “H, subscript 2, S e” and is at point “4, negative 45.” The fourth point on the line is labeled “H, subscript 2, T e” and is at point “5, negative 3.”" style="zoom:80%;" />
Note:
In comparison to periods 3−5, the binary hydrides of period 2 elements in groups 17, 16 and 15 (F, O and N, respectively) exhibit anomalously high boiling points due to hydrogen bonding.
---
## Question 7
Define the following and give an example of each:
----
**Dispersion Force**
<p style="text-align:left; color:green;">Dispersion forces occur as an atom develops a temporary dipole moment when its electrons are distributed asymmetrically about the nucleus. This structure is more prevalent in large atoms such as argon or radon. A second atom can then be distorted by the appearance of the dipole in the first atom. The electrons of the second atom are attracted toward the positive end of the first atom, which sets up a dipole in the second atom. The net result is rapidly fluctuating, temporary dipoles that attract one another.</p>
<!-- .element: class="fragment" data-fragment-index="1" -->
----
**Dipole‑Dipole Attraction**
<p style="text-align:left; color:green;">A dipole-dipole attraction is a force that results from an electrostatic attraction of the positive end of one polar molecule for the negative end of another polar molecule.</p>
<!-- .element: class="fragment" data-fragment-index="1" -->
----
**Hydrogen Bond**
Solution
<p style="text-align:left; color:green;">Hydrogen bonds form whenever a hydrogen atom is bonded to one of the more electronegative atoms, such as a fluorine, oxygen, nitrogen, or chlorine atom. The electrostatic attraction between the partially positive hydrogen atom in one molecule and the partially negative atom in another molecule gives rise to a strong dipole-dipole interaction called a hydrogen bond</p>
<!-- .element: class="fragment" data-fragment-index="1" -->
---
## Problem 9
Why do the boiling points of the noble gases increase in the order $\ce{He < Ne < Ar < Kr < Xe}$?
<p style="color:green">The London forces typically increase as the number of electrons increase.</p>
<!-- .element: class="fragment" data-fragment-index="1" -->
---
## Problem 10
<p style="text-align:left">
Neon (T<sub>B</sub> = -245.9 °C) and HF (T<sub>B</sub> = -83.1 °C) have approximately the same molecular masses.
</p>
<br/>
<p style="text-align:left">
a. Explain why the boiling points of Neon and HF differ.
</p>
<p style="text-align:left">
b. Compare the change in the boiling points of Ne, Ar, Kr, and Xe with the change of the boiling points of HF, HCl, HBr, and HI, and explain the difference between the changes with increasing atomic or molecular mass.
</p>
----
a. The boiling point of Ne is –245.9 °C, whereas that of HF is –83.1 °C. As their molecular masses are similar, the higher boiling point of HF can be explained on the basis of its hydrogen bonds preventing the escape of molecules from the liquid surface.
b. In each case (HF and Ne; HCl and Ar; HBr and Kr; HI and Xe), the corresponding pairs have similar molecular or atomic masses. The much higher boiling point of HF compared with Ne is due to hydrogen bonding in HF. HCl has a much higher (about 100 °C) boiling point compared to Ar because of dipole-dipole interactions. Similarly, HBr and HI, cannot form hydrogen bonds, dipole-dipole interactions make the boiling points of HBr and HI approximately 60 °C to 40 °C higher than those of the corresponding atomic species.
---
## Problem 17
<p style="text-align:left">
Explain why a hydrogen bond between two water molecules is weaker than a hydrogen bond between two hydrogen fluoride molecules.
</p>
---
## Problem 18
<p style="text-align:left">
Under certain conditions, molecules of acetic acid, CH<sub>3</sub>COOH, form “dimers,” pairs of acetic acid molecules held together by strong intermolecular attractions:
</p>
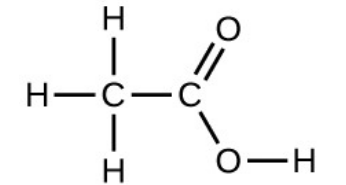
<p style="text-align:left">
Draw a dimer of acetic acid, showing how two CH<sub>3</sub>COOH molecules are held together, and stating the type of IMF that is responsible
</p>
----
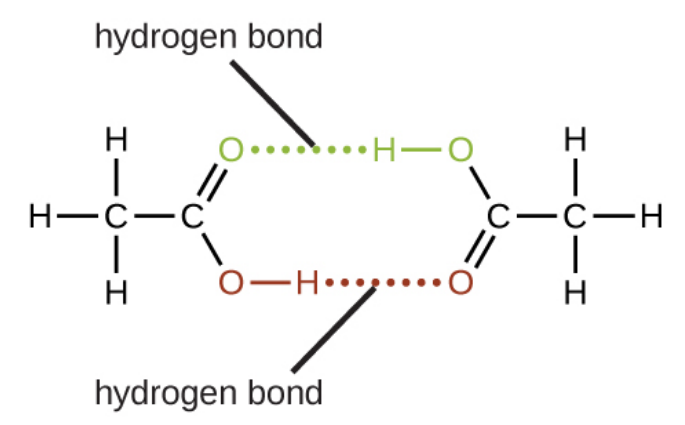
---
## Problem 20
<p style="text-align:left">
The density of liquid NH<sub>3</sub> is 0.64 g/ml; the density of gaseous NH<sub>3</sub> at STP is 0.0007 g/ml. Explain the difference between the densities of these two phases.
</p>
----
Molecules of gases are widely separated, whereas molecules in a liquid touch. Thus, a given volume of a gas contains a much smaller number of molecules and a much smaller mass than the same volume of a liquid.