<style>
/* Width of text in slides */
.slides { width: 1200px !important; }
/* line space bullet list */
.reveal .slides section, .reveal .slides section > section { line-height: 1.5; }
/* headers */
.reveal h1 {
margin-bottom: 1em;
font-weight: bold;
font-size: 32pt }
.reveal h2 {
margin-bottom: 1em;
font-weight: bold;
font-size: 30pt
}
.reveal h3 {
margin-bottom: 1em;
font-weight: bold;
font-size: 28pt
}
.reveal h4 {
margin-bottom: 1em;
font-weight: bold;
font-size: 26pt
}
.reveal h5 {
margin-bottom: 1em;
font-weight: bold;
font-size: 24pt
}
.reveal h6 {
margin-bottom: 1em;
font-weight: normal;
font-size: 24pt
}
/* text */
.reveal {
font-size: 24pt;
font-weight: normal;
}
/* logo as background */
.container {
background: no-repeat url(https://www.mcc.edu/MottStrong/images/mott-strong-3c-logo.png) top 20px left 10px; background-size: 5% !important;
} code {color: #000000}
.reveal table th { font-size: 0.80em}
.reveal table td { font-size: 0.70em}
/* Remove Border arround images */
.reveal section img {
background:none;
border:none;
box-shadow:none;
object-fit: contain;
margin: 0;
padding: 0;
max-width: 100%;
}
.reveal figure img {
box-shadow:none;
background:none;
border:none;
max-width: 100%;
object-fit: contain;
}
</style>
$\require{mhchem}$
$\require{cancel}$
OpenStax Chemistry 2e Chapter 3 Section 4
===
Michael Stogsdill
Mott Community College
---
## Learning Objectives
- Define the concentration units of mass percentage, volume percentage, mass-volume percentage, parts-per-million (ppm), and parts-per-billion (ppb)
- Perform computations relating a solution’s concentration and its components’ volumes and/or masses using these units
---
## Mass Percentage
- **Mass percentage** ($\%$ or $\% \textsf{mass}$) is a concentration defined as the mass of a component divided by the total mass, expressed as a percentage.
- The *mass percentage* of a *solute* is defined as the ratio of the *solute’s mass* to the *solution’s mass*, expressed as a percentage.
<br/>
$$
\%\text{mass} = \frac{\text{Component Mass}}{\text{Total Mass}} \times 100\% = \frac{\text{Solute Mass}}{\text{Solution Mass}} \times 100\%
$$
---
- Mass percentage is also called percent mass, percent weight ($\textsf{wt}\%$ or $\%\textsf{wt}$), weight over weight percent ($(\textsf{w/w})\%$), mass over mass percent ($\textsf{m/m}\%$) and other variations on this theme.
- The solute and solvent mass *must have the same units*. Though *it does not matter which mass units are used*. They will ultimately cancel.
---
<img src="https://openstax.org/apps/archive/20250522.165258/resources/303ba526a243c9bcedd4c1221b5748f9305365b9" alt="The sides of two cylindrical containers are shown. Each container’s label is partially visible. The left container’s label reads “Bleach.” The right label contains more information about the product including the phrase, “Contains: Sodium hypochlorite 7.4 %.”" style="zoom:70%" />
Note:
[**Figure 3.17**](https://openstax.org/books/chemistry-2e/pages/3-4-other-units-for-solution-concentrations#CNX_Chem_03_05_bleach) Liquid bleach is an aqueous solution of sodium hypochlorite ($\ce{NaOCl}$). This brand has a concentration of 7.4% $\ce{NaOCl}$ by mass.
---
## Question 69
Consider this question: What mass of a concentrated solution of nitric acid (68.0% HNO3 by mass) is needed to prepare 400.0 g of a 10.0% solution of HNO3 by mass?
----
Outline the steps necessary to answer the question.
<br/>
This is a straight forward dilution problem.
<!-- .element: class="fragment" data-fragment-index="1" -->
<br/>
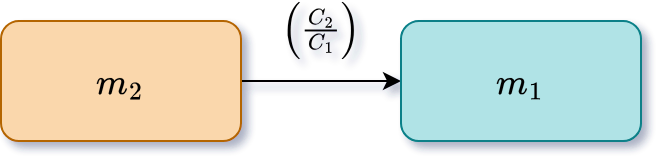
<!-- .element: class="fragment" data-fragment-index="2" -->
----
Answer the question.
<br/>
$C_1 = 68.0 \%$
<!-- .element: class="fragment" data-fragment-index="1" -->
$C_2 = 10.0 \%$
<!-- .element: class="fragment" data-fragment-index="2" -->
$m_2 = 400.0 \, \textsf{g}$
<!-- .element: class="fragment" data-fragment-index="3" -->
<br/>
$C_1 m_1 = C_2 m_2$
<!-- .element: class="fragment" data-fragment-index="4" -->
$m_1 = m_2 \left( \displaystyle\frac{C_2}{C_1} \right) = 400.0 \, \textsf{g} \left( \displaystyle\frac{10.0 \%}{68.0 \%} \right) = 58.8 \, \textsf{g}$
<!-- .element: class="fragment" data-fragment-index="5" -->
---
## Question 70
What mass of a 4.00% $\ce{NaOH}$ solution by mass contains 15.0 g of NaOH?
----
Do yourself a favor. When you see 4.00% $\ce{NaOH}$, think
<br/>
$$
\frac{4.00 \, \textsf{g} \, \ce{NaOH}}{100.00 \, \textsf{g} \, \textsf{solution}}
$$
<br/>
From here, we can reason that 200 g of solution would contain 8.00 g $\ce{NaOH}$, 300 g of solution would contain 12.00 g of $\ce{NaOH}$, and so on.
<!-- .element: class="fragment" data-fragment-index="1" -->
----
To solve for the mass of solution containing 15 g we divide by the concentraton.
<br/>
$15.0 \, \textsf{g} \, \ce{NaOH} \left( \displaystyle\frac{100.00 \, \textsf{g} \, \textsf{solution}}{4.00 \, \textsf{g} \, \ce{NaOH}} \right) = 375 \, \textsf{g} \, \textsf{solution}$
<!-- .element: class="fragment" data-fragment-index="1" -->
---
## Question 71
What mass of solid $\ce{NaOH}$ (97.0% NaOH by mass) is required to prepare 1.00 L of a 10.0% solution of NaOH by mass? The density of the 10.0% solution is 1.109 g/mL.
----
A 10.0% solution contains 10.0 g of $\ce{NaOH}$ for every 100 g of solution.
$$
\frac{10.0 \, \textsf{g} \, \ce{NaOH}}{100.0 \, \textsf{g solution} }
$$
<!-- .element: class="fragment" data-fragment-index="1" -->
<br/>
The mass of $\ce{NaOH}$ per mL of solution can be calculated by multiplying by the density
<!-- .element: class="fragment" data-fragment-index="2" -->
$\left( \displaystyle\frac{10.0 \, \textsf{g} \, \ce{NaOH}}{100.0 \, \textsf{g solution}} \right) \left( \displaystyle\frac{1.109 \, \textsf{g solution}}{ 1.000 \, \textsf{mL solution}} \right) = \displaystyle\frac{ 0.111 \, \textsf{g} \, \ce{NaOH}}{1.00 \, \textsf{mL solution}}$
<!-- .element: class="fragment" data-fragment-index="3" -->
----
We can now calculate the mass of sodium hydroxide by multiplying by the volume. We must match the units of volume.
$1000 \, \textsf{mL} \left( \displaystyle\frac{ 0.111 \, \textsf{g} \, \ce{NaOH}}{1.00 \, \textsf{mL solution}} \right) = 111 \, \textsf{g} \, \ce{NaOH}$
<!-- .element: class="fragment" data-fragment-index="1" -->
<br/>
But wait :exclamation: There is still a problem :frowning:. The "stuff" we have in our jar is only 97.0% $\ce{NaOH}$. We need to account for this.
<!-- .element: class="fragment" data-fragment-index="2" -->
$111 \, \textsf{g} \, \ce{NaOH} \left( \displaystyle\frac{ 100 \, \textsf{g stuff}}{97.0 \, \textsf{g} \, \ce{NaOH}} \right) = 114 \, \textsf{g stuff in the jar}$
<!-- .element: class="fragment" data-fragment-index="3" -->
---
## Question 72
What mass of $\ce{HCl}$ is contained in 45.0 mL of an aqueous $\ce{HCl}$ solution that has a density of 1.19 g cm^–3^ and contains 37.21% $\ce{HCl}$ by mass?
----
First, we will calculate the mass per milliter of solution.
$\left( \displaystyle\frac{37.21 \, \textsf{g} \, \ce{HCl}}{100 \, \textsf{g solution}} \right) \left( \displaystyle\frac{1.19 \, \textsf{g solution}}{1.00 \, \textsf{mL solution}} \right) = \displaystyle\frac{0.443 \, \textsf{g} \, \ce{HCl}}{1.00 \, \textsf{mL solution}}$
<!-- .element: class="fragment" data-fragment-index="1" -->
<br/>
Now, we can calulate the mass in 45.0 mL.
<!-- .element: class="fragment" data-fragment-index="2" -->
$45.0 \, \textsf{mL} \left( \displaystyle\frac{0.443 \, \textsf{g} \, \ce{HCl}}{1.00 \, \textsf{mL solution}} \right) = 19.9 \, \textsf{g} \, \ce{HCl}$
<!-- .element: class="fragment" data-fragment-index="3" -->
----
We can combine these calculations into one expression like this
<br/>
$45.0 \, \textsf{mL} \left( \displaystyle\frac{37.21 \, \textsf{g} \, \ce{HCl}}{100 \, \textsf{g solution}} \right) \left( \displaystyle\frac{1.19 \, \textsf{g solution}}{1.00 \, \textsf{mL solution}} \right) = 19.9 \, \textsf{g} \, \ce{HCl}$
---
## Question 77
Copper(I) iodide ($\ce{CuI}$)is often added to table salt as a dietary source of iodine. How many moles of $\ce{CuI}$ are contained in 1.00 lb (454 g) of table salt containing 0.0100% $\ce{CuI}$ by mass?
----
First, we'll calculate the mass of $\ce{CuI}$ in 454 g of salt.
$454 \, \textsf{g salt} \left( \displaystyle\frac{0.01 \, \textsf{g} \, \ce{CuI}}{100 \, \textsf{g salt}} \right) = 0.0454 \, \textsf{g} \, \ce{CuI}$
<!-- .element: class="fragment" data-fragment-index="1" -->
<br/>
Now we convert this value to moles by dividing by the molar mass.
<!-- .element: class="fragment" data-fragment-index="2" -->
$0.0454 \, \textsf{g} \, \ce{CuI} \left( \displaystyle\frac{1 \, \textsf{mol}}{190.45 \, \textsf{g}} \right) = 2.38 \times 10^{-4} \, \textsf{mol} \, \ce{CuI}$
<!-- .element: class="fragment" data-fragment-index="3" -->
---
## Volume Percentage
- The concentration of a solution formed by dissolving a liquid solute in a liquid solvent is often expressed as a **volume percentage** ($\%\textsf{vol}$ or $(\textsf{v/v})\%$).
<br/>
$$
\%\text{vol} = \frac{\text{Solute Volume}}{\text{Solution Volume}} \times 100\%
$$
---
- The solute and solvent volumes *must have the same units*. Though *it does not matter which units are used*. They will ultimately cancel.
- Because volume is not a conserved quantity and the density of liquids can vary significantly with temperature, $\%\textsf{vol}$ can be an unreliable unit to use.
---
## Mass-Volume Percentage
- **Mass-volume percent** ($\textsf{(m/v)}\%$)is a ratio of a solute’s mass to the solution’s volume expressed as a percentage.
<br/>
$$
\text{(m/v)}\% = \frac{\text{Solute Mass}}{\text{Solution Volume}} \times 100\%
$$
---
- Unlike *mass percent* and *volume percent* the units will not cancel and definitely matter.
- The specific units used for solute mass and solution volume may vary between appplications and disciplines.
- Physiological saline solution, used to prepare intravenous fluids, has a concentration of 0.9 (m/v)%, indicating that the composition is 0.9 g of solute per 100 ml of solution.
- *Mass-volume percent* is often ambiguous, which is why I don’t use it.
- *Mass-volume percent* is also temperature dependent
---
<img src="https://openstax.org/apps/archive/20250522.165258/resources/4379f79d1444552d0754c1ce717385490108ac2a" alt="Two pictures are shown labeled a and b. Picture a depicts a clear, colorless solution in a plastic bag being held in a person’s hand. Picture b shows a person’s hand holding a detection meter with a digital readout screen while another hand holds someone’s finger up to the end of the meter. The meter is pressed to the drop of blood that is at the end of the person’s finger." style="zoom:80%" />
Note:
[**Figure 3.18**](https://openstax.org/books/chemistry-2e/pages/3-4-other-units-for-solution-concentrations#CNX_Chem_03_05_saline) “Mixed” mass-volume units are commonly encountered in medical settings. (a) The NaCl concentration of physiological saline is 0.9% (m/v). (b) This device measures glucose levels in a sample of blood. The normal range for glucose concentration in blood (fasting) is around 70–100 mg/dL. (credit a: modification of work by “The National Guard”/Flickr; credit b: modification of work by Biswarup Ganguly)
---
## Parts per Million and Parts per Billion
- Very low solute concentrations are often expressed using **parts per million** ($\textsf{ppm}$) or **parts per billion** ($\textsf{ppb}$).
<br/>
**ppm**
$$
\text{ppm} = \frac{\text{Solute Mass}}{\text{Solution Mass}} \times 10^6
$$
**ppb**
$$
\text{ppb} = \frac{\text{Solute Mass}}{\text{Solution Mass}} \times 10^9
$$
:::info
:point_right: Note that *ppm* is %mass times 10^4^ and *ppb* is %mass times 10^7^.
:::
---
<img src="https://openstax.org/apps/archive/20250522.165258/resources/dd02c0dab168431a8e695efdbd00906a7f6ba510" alt="Two pictures are shown labeled a and b. Picture a is a close-up shot of water coming out of a faucet. Picture b shows a machine with the words, “Filtered Water Dispenser.” This machine appears to be inside a refrigerator." style="zoom:80%" />
Note:
[**Figure 3.19**](https://openstax.org/books/chemistry-2e/pages/3-4-other-units-for-solution-concentrations#CNX_Chem_03_05_faucet) (a) In some areas, trace-level concentrations of contaminants can render unfiltered tap water unsafe for drinking and cooking. (b) Inline water filters reduce the concentration of solutes in tap water. (credit a: modification of work by Jenn Durfey; credit b: modification of work by “vastateparkstaff”/Wikimedia commons)
---
### Convenient approximations
- Parts per million can be approximated as
$$
\frac{\textsf{mg} \text{ of solute}}{\textsf{L}\text{ of solution}}
$$
- Parts per billion can be approximated as
$$
\frac{\mu\textsf{g} \text{ of solute}}{\textsf{L}\text{ of solution}}
$$
- These approximations hold true so long as the solution's density doesn’t deviated significantly from 1 g/mL.
- Most aqueous solutions will have a density close to 1 g/mL.
---
## Question 73
The hardness of water (hardness count) is usually expressed in parts per million (by mass) of $\ce{CaCO3}$, which is equivalent to milligrams of $\ce{CaCO3}$ per liter of water. What is the molar concentration of $\ce{Ca^2+}$ ions in a water sample with a hardness count of 175 mg $\ce{CaCO3}$/L?
----
We know that 1 liter of water contains 175 mg of $\ce{CaCO3}$. We just need to figure out how many moles this is by dividing by the molar mass.
$n = 0.175 \, \textsf{g} \left( \displaystyle\frac{1 \, \textsf{mol}}{100.09 \, \textsf{g}} \right) = 1.75 \times 10^{-3} \, \textsf{mol} \, \ce{CaCO3}$
<!-- .element: class="fragment" data-fragment-index="1" -->
<br/>
Since this is the number of moles contained in 1 liter, it is numerically equaivalent to the molarity.
<!-- .element: class="fragment" data-fragment-index="2" -->
$C_M = \displaystyle\frac{n}{V} = \displaystyle\frac{1.75 \times 10^{-3} \, \textsf{mol}}{1 \, \textsf{L}} = 1.75 \times 10^{-3} \, \textsf{M}$
<!-- .element: class="fragment" data-fragment-index="3" -->
---
## Question 74
The level of mercury in a stream was suspected to be above the minimum considered safe (1 part per billion by weight). An analysis indicated that the concentration was 0.68 parts per billion. Assume a density of 1.0 g/mL and calculate the molarity of mercury in the stream.
----
Let's see why our approximation converges to the true value when the density of a solution is one.
<br/>
$0.68 \, \textsf{ppm} = \displaystyle\frac{0.68 \times 10^{-9} \, \textsf{g} }{ 1.0 \, \textsf{g water} } \left( \displaystyle\frac{ 1.0 \, \textsf{g water} }{ 1.0 \, \textsf{mL water} } \right) = \displaystyle\frac{0.68 \times 10^{-9} \, \textsf{g} }{ 1.0 \, \textsf{mL water} } = \displaystyle\frac{0.68 \, \textsf{ng}}{1.0 \, \textsf{mL} }$
<!-- .element: class="fragment" data-fragment-index="1" -->
$\displaystyle\frac{0.68 \, \textsf{ng}}{1.0 \, \textsf{mL} } \left( \displaystyle\frac{ 1000 \, \textsf{mL} }{ 1 \, \textsf{L} } \right) = \displaystyle\frac{ 680 \, \textsf{ng} }{ 1.0 \, \textsf{L} } = \displaystyle\frac{ 0.68 \, \mu \textsf{g} }{ 1.0 \, \textsf{L} }$
<!-- .element: class="fragment" data-fragment-index="2" -->
<br/>
You can see why I prefer to skip this derivation everytime I use ppb and instead think of it as an approximation.
<!-- .element: class="fragment" data-fragment-index="3" -->
----
Now, we just need to calculate how many moles is in 0.68 $\mu \textsf{g}$ of mercury.
<br/>
$\displaystyle\frac{ 0.68 \, \mu \textsf{g} }{ 1.0 \, \textsf{L} } \left( \displaystyle\frac{ 1 \, \textsf{g} }{ 1 \times 10^{6} \, \mu \textsf{g} } \right) \left( \displaystyle\frac{ 1 \, \textsf{mole} }{ 200.59 \, \textsf{g} } \right) = 3.4 \times 10^{-9} \, \textsf{M}$
<!-- .element: class="fragment" data-fragment-index="1" -->
---
## Question 75
In Canada and the United Kingdom, devices that measure blood glucose levels provide a reading in millimoles per liter. If a measurement of 5.3 mM is observed, what is the concentration of glucose ($\ce{C6H12O6}$) in mg/dL?
----
Let's tackle this one as one large expression
<br/>
$$
\frac{5.3 \, \textsf{mmol}}{1 \, \textsf{L}} \left( \frac{1 \, \textsf{mol}}{1000 \, \textsf{mmol}} \right) \left( \frac{ 180.16 \, \textsf{g}}{ 1 \, \textsf{mol}} \right) \left( \frac{ 1000 \, \textsf{mg}}{ 1 \, \textsf{g}} \right) \left( \frac{ 1 \, \textsf{L}}{ 10 \, \textsf{dL}} \right) = 95 \, \frac{\textsf{mg}}{\textsf{dL}}
$$
{"title":"OpenStax Chemistry 2e Chapter 3 Section 4","type":"slide","tags":"Chemistry 2e, CHEM 131, Chapter 3","slideOptions":{"theme":"white","transition":"slide","controls":true,"progress":true,"defaultTiming":120,"slideNumber":false,"history":false,"keyboard":true,"overview":true,"center":true,"touch":true,"loop":false,"rtl":false,"shuffle":false,"fragments":true,"embedded":false,"help":true,"showNotes":false,"autoPlayMedia":null,"autoSlide":0,"autoSlideStoppable":true,"autoSlideMethod":"Reveal.navigateNext","mouseWheel":false,"hideAddressBar":true,"previewLinks":false,"transitionSpeed":"default","backgroundTransition":"fade","viewDistance":3}}