$\require{mhchem}$
Eudiometry
===
## Introduction
A eudiometer is an analytical apparatus used to trap gases that are generated during a chemical reaction. In analytical chemistry, gases generated in chemical reactions can be collected to investigate the properties of a substance, determine the percent yield of reaction and provide insight into the chemical composition of an unknown. In this experiment, we will use eudiometry to calculate the percent yield of the neutralization reaction between calcium carbonate and hydrochloric acid. We will then use the percent yield to determine the mass composition of a calcium carbonate and sodium chloride mixture of unknown composition.
### Determining Percent Yield
We will be investigating the reaction between calcium carbonate and hydrochloric acid.
$$
\ce{CaCO3 ($s$) + 2HCl ($aq$) -> CaCl2 ($aq$) + CO2 ($g$) + H2O ($l$)}
$$
We can see from the balanced equation that the reaction produces carbon dioxide ($\ce{CO2}$) gas. By measuring the temperature, pressure, and volume of the gas, we can determine the number of moles of carbon dioxide gas produced assuming it behaves as an ideal gas.
$$
\large n_{\ce{CO2}} = \normalsize \frac{PV}{RT} \hspace{10mm} \text{Eqn. 1}
$$
We can then multiply the number of moles of carbon dioxide by the
appropriate stoichiometric ratio to find the number of moles of calcium carbonate that has reacted.
$$
\large n_{\ce{CO2}} \normalsize \left( \frac{1 \, \textsf{mole} \, \ce{CaCO3}}{1 \, \textsf{mole} \, \ce{CO2}} \right) = \large n^\textsf{reacted}_{\ce{CaCO3}} \hspace{10mm} \normalsize \text{Eqn. 2}
$$
When we ensure there is an excess of hydrochloric acid, we can calculate the actual number of moles of calcium carbonate based on the mass we initially weigh out. Dividing the mass by the formula mass of calcium carbonate (100.087 $\ce{\frac{g}{mole}}$ ) we get the theoretical moles of calcium carbonate.
$$
\large \frac{m_{\ce{CaCO3}}}{M^w} = n^\textsf{actual}_{\ce{CaCO3}} \hspace{10mm} \normalsize \text{Eqn. 3}
$$
The percent yield is simply the ratio of the actual yield to the theoretical yield times one hundred percent.
$$
\% \text{Yield} = \large \left( \frac{n^\textsf{reacted}_{\ce{CaCO3}}}{n^\textsf{actual}_{\ce{CaCO3}}} \right) \normalsize \times 100\% \hspace{10mm} \text{Eqn. 4}
$$
### Measuring Temperature, Pressure, and Volume
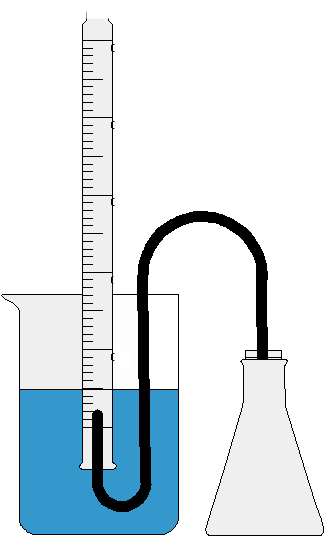
**Figure 1:** Eudiometer apparatus
You will prepare your eudiometry set up by first filling a gas collection tube with water. You'll then fill a large beaker half way with water. Holding your finger over the mouth of the tube, you'll invert it and place it under the water level in the container. Clamp the tube in place and remove your finger. The water will remain inside the tube because no air can enter to replace the displaced water. You'll then need to prepare the chamber where the actual reaction will take place. This is a vacuum flask fitted with a stopper. Route the tubing into the gas collection tube in such a way that any gases will be released into it.
To measure the temperature of the carbon dioxide $\left( T_{\ce{CO_2}} \right)$, we will assume it has come to equilibrium with the water. This is reasonable considering that the gas bubbles will need to pass through the water making good thermal contact.
Measuring the volume of the gas is straight forward. You will simply measure the final level of the water from the bottom of the meniscus, like you would with a buret.
Determining the pressure of the gas captured in the eudiometer is not as straight forward as the temperature and volume. The water in the container is open to the atmosphere. The pressure of the column of gas is equal to atmospheric pressure. However, not all of the gas columns pressure is due to the pressure of carbon dioxide $\left( P_{\ce{CO_2}} \, \right)$. Some of the pressure is due to the water vapor $\left( P_\textsf{water} \hspace{1.5mm} \right)$ and the hydrostatic pressure of the water column $\left( P_\textsf{column} \hspace{2mm} \right)$. The relationship between all of these quantities can be expressed using Dalton's Law.
$$
\large P_\textsf{atm} = P_{\ce{CO_2}} + P_\textsf{water} + P_\textsf{column} \normalsize \hspace{10mm} \text{Eqn. 5}
$$
The hydrostatic water pressure $\left( P_\textsf{column} \hspace{2mm} \right)$ is equal to the mass of the water ($m$) times the acceleration of gravity $\left( g, \, 9.8 \displaystyle\frac{\text{m}}{\text{s}^2} \right)$ divided by the area of the water column in contact with the gas column ($A$).
$$
P_\textsf{column} \hspace{1.5mm} = \frac{mg}{A} \hspace{10mm} \text{Eqn. 6}
$$
We don't know the mass of the water but we can measure its volume
$\left( V_\textsf{water} \hspace{1.5mm} \right)$ just as we did with the gas column. We can then multiply the volume by the density of the water ($\rho$) and substitute the quantity into equation 6. The value of $\rho$ can be determined using the [Density of Water at Different Temperatures](https://doc.chemnotes.org/s/waterdensity#Density-of-Water-at-Different-Temperatures) table.
$$
P_\textsf{column} \hspace{1.5mm} = \frac{\rho V_\textsf{water} \hspace{2mm} g}{A} \hspace{10mm} \text{Eqn. 7}
$$
Rewriting the volume of water and area in terms of the radius of the eudiometer and height of the column of water $\left( P_\textsf{water} \hspace{1.5mm} \right)$ and then simplifying the result. we arrive at a simple equation for $\left( P_\textsf{column} \hspace{2mm} \right)$.
$$
P_\textsf{column} \hspace{1.5mm} = \frac{\rho \pi r^2 h_\textsf{water} \hspace{2mm} g}{\pi r^2} = \rho h_\textsf{water} \hspace{2mm} g \hspace{10mm} \text{Eqn. 8}
$$
The vapor pressure of the water $\left( P_\textsf{water} \hspace{1.5mm} \right)$ depends only on the temperature of the water and is well documented. Table 1 contains the vapor pressure of water at various temperatures.
| Temperature | Vapor Pressure (torr) | Vapor Pressure (Pa) |
| :---------: | :-------------------: | :-----------------: |
| 0 | 4.6 | 613.2812 |
| 4 | 6.1 | 813.2642 |
| 10 | 9.2 | 1226.562 |
| 15 | 12.8 | 1706.522 |
| 20 | 17.5 | 2333.135 |
| 22 | 19.8 | 2639.776 |
| 25 | 23.8 | 3173.064 |
| 30 | 31.8 | 4239.64 |
| 35 | 42.2 | 5626.188 |
| 40 | 55.3 | 7372.707 |
| 45 | 71.9 | 9585.852 |
| 50 | 92.5 | 12332.29 |
| 55 | 118.0 | 15732 |
| 60 | 149.4 | 19918.31 |
| 65 | 187.5 | 24997.88 |
| 70 | 233.7 | 31157.35 |
| 75 | 289.1 | 38543.39 |
| 80 | 355.1 | 47342.64 |
| 85 | 433.6 | 57808.42 |
| 90 | 525.8 | 70100.71 |
| 95 | 633.9 | 84512.82 |
| 100 | 760.0 | 101324.7 |
**Table 1**
We can simply look up the value closest to the water temperature we have already measured to find this correction. Once we have determined the atmospheric pressure, hydrostatic pressure and water vapor , we can finally calculate the pressure of carbon dioxide by rearranging equation 5.
$$
P_{\ce{CO_2}} = P_\textsf{atm} - P_\textsf{water} \, - P_\textsf{column} \hspace{10mm} \text{Eqn. 9}
$$
### Sample Problem
A student conducts a eudiometer experiment. They determine that the temperature of the water was 27.1 °C and the height of water column was 41.6 cm. The atmospheric pressure in the room was 100.1 kPa. What is pressure of carbon dioxide in the gas collection tube?
The units are very important in this problem The first thing to keep in mind is that a pascal is a derived unit equal to kilograms per meter per second squared.
$$
\ce{
Pa = \frac{kg}{m \cdot s^2}
}
$$
We will need to convert the values given to be consistent with the units of pascals. The first thing we will need to do is look up the density of water at 27.1 °C in table 2.
$$
\rho = 0.996512 \, \frac{\textsf{g}}{\textsf{ml}} \left( \frac{1000 \, \frac{\textsf{kg}}{\textsf{m}^3}}{1 \, \frac{\textsf{g}}{\textsf{ml}}} \right) = 996.512 \, \frac{\textsf{kg}}{\textsf{m}^3}
$$
Next we will need to convert the height of the water column to meters.
$$
h_\textsf{water} \, = 41.6 \, \textsf{cm} \left( \frac{1 \, \textsf{m}}{100 \, \textsf{cm}} \right) = 0.416 \, \textsf{m}
$$
We can now calculate the hydrostatic pressure in the column.
$$
P_\textsf{column} \hspace{2mm} = \rho h_\textsf{water} \hspace{2mm} g = \left(996.512 \, \frac{\textsf{kg}}{\textsf{m}^3} \right) \left( 0.416 \, \textsf{m} \right) \left( 9.8 \, \frac{\textsf{m}}{\textsf{s}^2} \right) = 4060 \, \frac{\textsf{kg}}{\textsf{m} \cdot \textsf{s}^2} = 4060 \, \textsf{Pa} = 4.060 \, \textsf{kPa}
$$
Next, we will look up the vapor pressure of water at this temperature and convert it to kPa.
$$
P_\textsf{water} \, = 26.7 \, \textsf{torr} \left( \frac{101.4 \, \textsf{kPa}}{760 \, \textsf{torr}} \right) = 3.56 \, \textsf{kPa}
$$
Now we can calculate the pressure of carbon dioxide in the gas collection tube.
$$
P_{\ce{CO_2}} = 100.1 \, \textsf{kPa} - 3.56 \, \textsf{kPa} - 4.060 \, \textsf{kPa} = 92.5 \, \textsf{kPa}
$$
### Determining the Mass Composition of an Unknown
In the final part of our experiment, we will repeat our measurements with an unknown calcium carbonate and salt mixture. Once we determine the number of moles of calcium carbonate that has reacted in this experiment $\left( \large n^\textsf{reacted}_{\ce{CaCO_3}} \right)$, we can calculate the actual number of moles of calcium carbonate in the mixture by dividing by the %Yield as a decimal.
$$
\large \left( \frac{n^\textsf{reacted}_{\ce{CaCO3}}}{\% \text{Yield}} \right) 100\% = n^\textsf{reacted}_{\ce{CaCO3}} \left( \frac{n^\textsf{actual}_{\ce{CaCO3}}}{n^\textsf{reacted}_{\ce{CaCO3}}} \right) = n^\textsf{actual}_{\ce{CaCO3}} \normalsize \hspace{10mm} \text{Eqn. 10}
$$
The mass of calcium carbonate in the mixture can be calculated by multiplying the actual number of moles by the formula mass of calcium carbonate.
$$
\large n^\textsf{actual}_{\ce{CaCO3}} \left( M^{w} \right) = m_{\ce{CaCO3}} \normalsize \hspace{10mm} \text{Eqn. 11}
$$
Finally, we can calculate the %mass of calcium carbonate in our sample by dividing the mass calcium carbonate by the total mass of the sample.
$$
\% \text{mass} = \large \left( \frac{m_{\ce{CaCO3}}}{m_\textsf{total}} \right) \normalsize 100 \% \hspace{10mm} \text{Eqn. 12}
$$
## Materials
- Gas Collection Tube
- Large Beaker
- Tubing
- Stopper
- 1 M hydrochloric Acid
- Calcium Carbonate
- Calcium Carbonate of Unknown Purity
## Procedure
### Part A: Pure Calcium Carbonate
1. Prepare your eudiometer apparatus as it appears in figure 1. Don’t forget to add your Alka Seltzer tab.
2. Weigh out at least 0.100 g of calcium carbonate and add it to your gel cap.
3. Take your flask back to your station and test your stopper to make sure it fits snugly. Any gas that escapes during the reaction will effect your results.
4. Obtain 10 ml of 1 M hydrochloric acid in a graduated cylinder.
5. Add the hydrochloric acid to the flask and replace the stopper quickly. Once your gel cap has dissolved, you should see a vigorous reaction begin.
6. Gently swirl the flask to ensure a complete reaction. Note that the gas is in fact being routed into your gas collection tube.
7. Once the reaction is complete and no more gas is evolved, measure the volume of gas evolved by reading the graduations on the gas collection tube.
8. Measure the height of the water column with your meter stick. Record this value on your data sheet.
9. Measure the temperature of the water with your thermometer. Record the temperature on your data sheet.
10. Repeat steps 1 - 8 two more times.
### Part B: Unknown Sodium Chloride Calcium Carbonate Mixture
1. Repeat the procedure in Part A three times with the unknown mixture. Be sure to record all your values on your data sheet.
<iframe width="100%" height="410" src="https://www.youtube.com/embed/bIwfe7VVmek" title="Chemnotes.org" frameborder="0" allow="accelerometer; autoplay; clipboard-write; encrypted-media; gyroscope; picture-in-picture; web-share" referrerpolicy="strict-origin-when-cross-origin" allowfullscreen></iframe>
## Data Sheet
{%pdf https://public.chemnotes.org/lab/sheets/eudiometry.pdf %}
<iframe width="100%" height="410" src="https://www.youtube.com/embed/Y1N8BR8qC6Y" title="Chemnotes.org" frameborder="0" allow="accelerometer; autoplay; clipboard-write; encrypted-media; gyroscope; picture-in-picture; web-share" referrerpolicy="strict-origin-when-cross-origin" allowfullscreen></iframe>